|
|
Detailed Explanation of Discrepant Event
|
|
|
- As the water is heated and boiled, steam displaces the air in the container above the water. When the soda can is plunged into cold water, the vapor condenses quickly, leaving a vacuum in most of the can. The resulting large discrepancy between the outside and inside air pressure leads to a large net inward force on the can, ending with its rapid crushing.
- The one gallon can operates similarly, but its capping and the fact that it is not plunged into cold water results in a slower condensation of the moisture inside the can. The one gallon can is much sturdier but atmospheric pressure is such that not even this container can withstand its forces. The can collapses as a vacuum is formed.
- The process can be viewed four different ways...
- The can has air pressure exerted on it both from outside and inside and those forces are responsible for the can's subsequent demise.
- Newton's second law deals with the sum of the forces. As long as the forces inside and outside are balanced and equal, the can undergoes no change. When the forces are unbalanced, changes in the shape of the metal container can and will take place.
- Pressure in a container is the sum of the partial pressures of the constituent gases. If one gas is removed (water vapor condensing) the total pressure of the gases in the container declines.
- As the container collapses, the volume of the container decreases. The pressure of a gas is related to the volume of its container. At some point the diminished volume results in an adequate pressure inside the container to prevent continued collapse.
|
|
Standards
H.S. Physics
1b. Students know that when forces are balanced, no acceleration occurs; thus an object continues to move at a constant speed or stays at rest (Newton’s first law).
1c. Students know how to apply the law F=ma to solve one-dimensional motion problems that involve constant forces (Newton’s second law).
H.S. Chemistry
4c. Students know how to apply the gas laws to relations between the pressure, temperature,
and volume of any amount of an ideal gas or any mixture of ideal gases.
4i.* Students know how to apply Dalton’s law of partial pressures to describe the composition of gases and Graham’s law to predict diffusion of gases.
|
|
Questioning Script
Prior knowledge & experience:
- Students know that an aluminum can can be crushed by hand but stronger cans such as a one gallon can, cannot.
- Students know that as one dives deep into a pool, they feel pressure against their ears.
- Steam can return to a liquid when cooled.
Root question:
- Does atmospheric pressure exert enough force to crush cans, even strong ones?
Target response:
- Can students relate air pressure (14.7 lbs/in2 ) to a force when applied over an area?
Common Misconceptions:
- Since students don't experience pressure's effects often they overlook it.
- The quantitative value of air pressure, 14.7 lbs/in, seems small and unable to have an effect.
- Steam is not a gas.
- Gases exert little pressure.
|
|
|
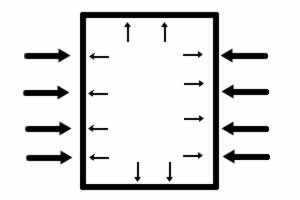
As the water boils, the pressure inside and outside the can are the same size. |
|
|
When the can is plunged into cold water, the water vapor condenses, leaving the pressure inside the can much less than outside the can. |
|
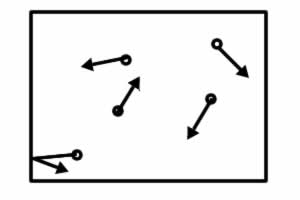
As the water boils, molecules bouncing off of the inside of the can create the forces pressing outward, these balance the atmosphere pressing inward. |
|
|
With water vapor condensing, fewer gas molecules are present, so fewer bounce off of the can and this leads to a smaller force pressing outward. |
|
References & Links:
Atmospheric Pressure (Wikipedia)
Kinetic Theory of Gases (NASA)
Newton's Laws (The Physics Classroom)
Gas Laws (Precision Measurement Equipment Lab)
Aluminum Beverage Can Story
Can Crushing (U. of Wisconsin)
Crushed Railroad Tank Car
|